CLE lecturer Jen Bernstein teamed up with researchers from Johns Hopkins University, MIT, and American University to publish an article in the newest issue of the Journal of the American Medical Informatics Association (JAMIA) examining collaboration in the research and development of pharmaceutical drugs.
In “Examining associations between cohesion and diversity in collaboration networks of pharmaceutical clinical trials with drug approvals,” the authors analyze the impact of diverse collaboration among nonprofit organizations, academic and government institutions, and pharmaceutical and biotech companies during the “incredibly complex and expensive process of drug development.”
Using data from Clinicaltrials.gov, the authors examined the characteristics of clinical trial networks and their impact on trial success, revealing clear benefits to collaboration diversity including “higher research output and efficiency.” Collaborations that lacked diversity were less successful in comparison.
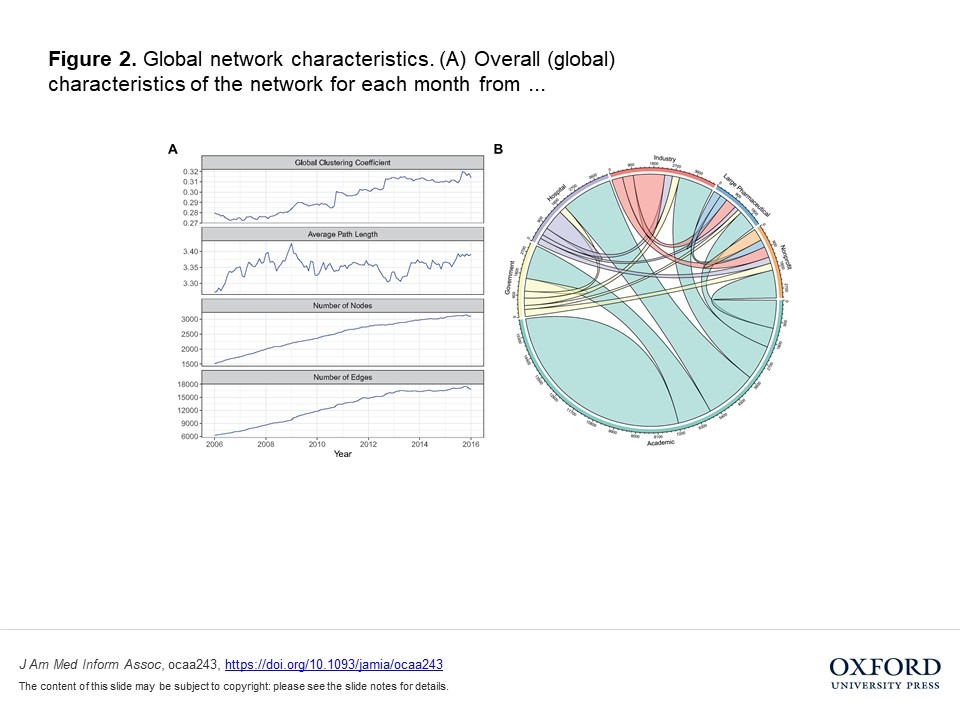
Bernstein says that the team’s research “contributes to the body of literature concerning a phenomenon known as ‘knowledge absorption,’ whereby collaborating with experts in different fields increases an actor’s knowledge base and ability to innovate by combining varying knowledge.” Though knowledge absorption in the pharmaceutical industry has been studied extensively by considering patents or contractual alliances, this study is the first to examine collaborative networks formed to run clinical trials.
While this project is now complete, three of Bernstein’s co-authors have been applying their analytical expertise to the COVID-19 pandemic. Dr. Gary Lin, lead author of this study and postdoctoral fellow with the Johns Hopkins Department of Emergency Medicine and the Center for Disease Dynamics, Economics & Policy (CDDEP) in Washington, D.C., works with his advisor Dr. Eili Klein to track and model COVID-19 data for the state of Maryland through the Johns Hopkins Emergency Medicine Internal COVID-19 Model. This model is one of three that Governor Larry Hogan consults regularly when making decisions for the state.
Also contributing to this study is Dr. Lauren Gardner, associate professor and co-director of the Center for Systems Science and Engineering at Johns Hopkins University. Named one of TIME’s 100 Most Influential People of 2020, Gardner created the free and open website, called the COVID-19 dashboard, widely used to track and visualize the COVID-19 pandemic in near real-time.
Diego Martinez, assistant professor of emergency medicine and health sciences informatics at the Johns Hopkins University School of Medicine. has also been tracking COVID-19 within the Johns Hopkins Health System. Martinez and co-authors have reported a variety of timely findings in journals including the Journal of the American Medical Association and the Journal of Hospital Infection.