
Researchers and clinicians don’t fully understand why some cancers spread and others do not. What they do know is that when cancer does spread, it dramatically decreases survival rates.
If physicians could predict the likelihood that primary tumors will metastasize, they would be able to choose the best treatment options for patients. However, current testing only reviews tumor genetics, which can mutate and change.
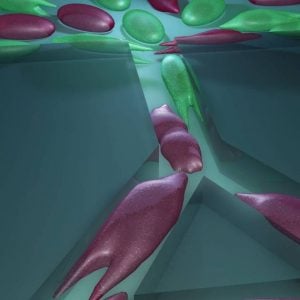
Breast cancer tumors contain a mixture of cells with different abilities, and the proportion of aggressive cells within a tumor determines its likelihood to metastasize. MAqCI identifies aggressive breast cancer cells (in purple) based on their ability to move from a feeder channel into narrower channels.
Chris Yankaskas, a PhD candidate in the Department of Chemical and Biomolecular Engineering at Johns Hopkins University, wondered if he could predict metastasis from a different angle, by instead looking at the cancer cell’s phenotype, or observable cell characteristics and behaviors. Under the direction of Konstantinos Konstantopoulos, a professor and core faculty member of the Institute for NanoBioTechnology, Yankaskas and a team of researchers created the Microfluidic Assay for Quantification of Cell Invasion, or MAqCI, a diagnostic tool and method for predicting breast cancer metastasis by looking at two key cell behaviors needed for metastasis instead of tumor genetics.
“The complexity of cancer progression and differences between each patient’s cancer cells make metastasis hard to predict on a case-by-case basis,” said Yankaskas. “We aim to continue working in breast cancer using cells from patients’ biopsies and hope to expand the technology to other cancer types.”