Johns Hopkins University scientists are part of a research team assessing how a person’s genetic profile affects the body. The results could help show how individual genetic differences contribute to disease and guide treatments for heritable disorders such as Alzheimer’s, high cholesterol, or Type 1 diabetes.
The human body has highly specialized organs and tissues that function very differently from each other, and each tissue is affected by different DNA sequencing. A variant is a spot in the genome where a person’s individual DNA sequence of four biochemicals—adenine, cytosine, guanine and thymine—is different from the standard human sequence.
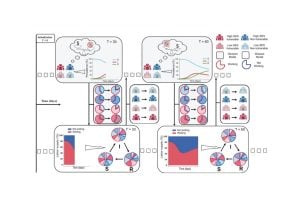
In two related articles published in the current edition of the journal Nature, the researchers report on the most expansive study ever conducted on how genetic patterns lead to molecular changes within specific tissues. One article shows results analyzing the genome-to-tissue link for genetic variants that are common in humans, and the other turns to the more challenging problem of predicting the effects of rare variants that are found in very few individuals, or even in just a single individual.
“There’s a lot we don’t understand about how genetic differences affect our bodies, and different diseases,” said Alexis Battle, a computer scientist in Johns Hopkins’ Whiting School of Engineering whose lab conducted the research analysis. She added that the work on rare genetic variants “is a step toward realizing the goal of personal genomics”—that is, when clinicians can identify the anomalies in an individual patient’s genetic code that cause diseases.
It is a challenging task. Of the 3 billion base pairs of the human genome—the biochemical “rungs” connecting the longer strands of the DNA molecule—scientists have only characterized the effects of a small percentage.
Part of the significance of the studies published in Nature lies in the size of the tissue sample, which is possible due to the work of the GTEx, or Genotype Tissue Expression project, an effort launched by the National Institutes of Health in 2010. The project has amassed and analyzed 7,051 tissue samples from 449 people.
In the studies, Battle and her colleagues used samples of 42 tissue types, including blood, liver, stomach, heart, brain, lung, and pancreas.
The scientists found tens of thousands of variants that have detectable effects in at least one of the tissues they studied, causing in some cases significant changes to levels of certain molecular markers, including proteins. The researchers also found 112 spots in the genome with long-range effects on molecular changes, including in tissues where these effects have not previously been detected. They also showed that for disease-associated variants known from other studies, they were able to detect molecular effects for approximately half, more than has ever been shown before.
“This is essential for understanding mechanisms of disease, and is only possible through the large number of tissues available in our dataset,” Battle said.
The scientists showed that genetic anomalies contribute significantly to extreme changes at the molecular level. Specifically, in 58 percent of cases where someone has extremely low levels of a chemical marker and in 28 percent of cases where they have extremely high levels, that person also harbors a rare genetic variant that could explain the molecular change.
The scientists also showed that their new analytical method provides a way to identify which rare genetic variants are likely to have an impact on the body. The method identifies such variants statistically significantly better than any existing methods, and could lead to better genetic diagnoses in clinical settings, Battle said.
Read more about Alexis Battle’s work in the Summer 2017 issue of JHU Engineering magazine.