Denis Wirtz is vice provost for research for Johns Hopkins University and the Theophilus Halley Smoot Professor in the Department of Chemical and Biomolecular Engineering at the Whiting School. He is also a member of the Data Science and AI Institute.
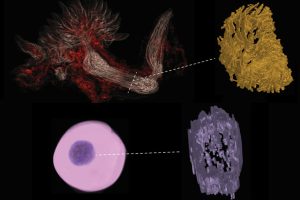
Through his research at the interface of physics, biology, and oncology, Wirtz has made seminal contributions to our understanding of cancer cell migration, cytoskeleton biophysics, and the nascent field of mechanobiology. He has developed quantitative methods, including particle-tracking microrheology, which are widely used both in academia and industry. He has also pioneered research in cell migration in 3D settings, bacterial cell division, and high-throughput cell phenotyping. Recently, he developed CODA, an AI-based method to image large volumes of tissues and tumors in 3D.
Wirtz co-founded the Johns Hopkins Institute for NanoBioTechnology (INBT) and is the director of the NCI-funded postdoctoral training program in nanotechnology for oncology, the NCI-funded Physical Sciences-Oncology Center (PS-OC), and the Johns Hopkins Cellular Cancer Biology Imaging Cancer (CCBIR) Center.
He is author and co-author of 250 peer-reviewed articles published in journals including Science, Nature, Cell, Nature Reviews Cancer, and Nature Cell Biology. His work at Johns Hopkins has been cited more than 31,000 times and has an h-index of 94.
Wirtz received the NSF CAREER award in 1995, was named Theophilus H. Smoot Professor of Engineering and Science in 2009, and was elected a Fellow of the Institute for Medical and Biological Engineering (AIMBE) in 2007, a Fellow of the American Association for the Advancement of Science (AAAS) in 2009, and a Fellow of the American Physical Society (APS) in 2010. He is also a member of the Royal Academy of Medicine of Belgium.
Wirtz received a physics engineering degree from the Free University of Brussels in 1988 and his MSc and PhD in chemical engineering from Stanford University in 1993. Wirtz has been the vice provost for research of Johns Hopkins University since 2014.