For a disease that’s disturbingly prevalent—an estimated 5.4 million people in the United States are affected—Alzheimer’s disease’s cause and cure remains elusive. But researchers know that the brain changes of Alzheimer’s start before symptoms such as memory loss show up. The same is true for other neurodegenerative diseases, such as Parkinson’s and Huntington’s disease.
“We need to be looking at the brain before someone starts to complain of significant clinical symptoms,” says Michael I. Miller, MS ’79, PhD ’84, the Herschel and Ruth Seder Professor of Biomedical Engineering and university Gilman Scholar.
Similar to his work to create a pediatric brain data bank, Miller is analyzing MRI brain images of the elderly. His goal: to unveil lurking signs of Alzheimer’s that cognitive tests cannot detect. He is collaborating with Marilyn S. Albert, director of the Division of Cognitive Neuroscience at the Johns Hopkins University School of Medicine, and Laurent Younes, chair of applied mathematics and statistics at the Whiting School.
Since 1995, Albert has been studying patients at risk for Alzheimer’s based on genetics and family history. One-fifth of 360 individuals have developed full-blown Alzheimer’s. Their brain scans are a critical window into the disease’s evolution.
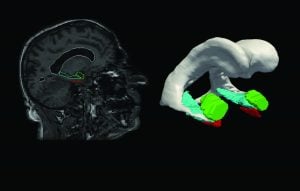
Now, using sophisticated computer models to study the shape and volume of three brain structures in the temporal lobe, associated with memory, the researchers have found significant changes in the entorhinal cortex 10 years before symptoms. Images show that the volume of gray matter shrinks, which is “consistent with losing neurons, which are the computing machinery associated with memory storage and retrieval,” says Miller.
The disease then moves to the hippocampus and the amygdala, which shrink to a smaller extent two to four years before symptom onset. Understanding these subtle brain changes could help design interventions that slow the disease. “If we find an important-enough-marker it could be used to diagnose and alter care,” Miller says.
Miller co-directs the Kavli Neuroscience Discovery Institute at Johns Hopkins with Richard Huganir, director of the Department of Neuroscience at the School of Medicine.




