Five years ago, Johns Hopkins neurosurgeon Nicholas Theodore brought a problem with echoes of the movie Groundhog Day to biomedical engineer Amir Manbachi, who was then teaching in the Undergraduate Design Team program at the Whiting School.
An expert in spinal cord injuries, Theodore was frank in noting that no significant new drugs or treatments had been developed over the previous half a century. In spinal cord injury repair, he told Manbachi and his students, surgeons are still doing “the same rudimentary things that we’ve been doing for 50 years.”
In the aftermath of an injury, they open up a patient and “decompress” the spinal cord, clearing out stray pieces of bone and pockets of hematoma to stabilize the spine. After surgery, they monitor blood pressure very carefully, tinkering frequently in an effort to reduce the risk of dangerous secondary injuries. Otherwise, however, the treatment regimen is mostly a matter of helping patients adjust to new realities marked by pain and paralysis.
- Amir Manbachi
- Nicholas Theodore
Theodore’s pitch to the Design Team program was that the way out of this Groundhog Day predicament might well be achieved through biomedical engineering. He wanted a team of Whiting School students to develop a map pointing the way toward better outcomes for spinal cord injury patients.
The pitch was a hit—one of the most popular proposals among the 20 or so options presented to students that year. In short order, Manbachi was supervising the work of a team headed by engineering student Ana Ainechi.
The map that emerged from that capstone project is still in play, though it has evolved over time into a highly ambitious undertaking. Late last year, Theodore and Manbachi received a five-year, $13.48 million grant from the Defense Advanced Research Projects Agency at the U.S. Department of Defense. Best known for its funding in the 1960s of ARPANET, a key precursor of the internet, DARPA specializes in supporting high-risk, high-reward projects.
This one aims to develop a one-two punch of cutting-edge devices with the potential to revolutionize both diagnostics and treatment in spinal cord injury cases.
That happy ending, however, will come to pass only if the team that Manbachi and Theodore have assembled can overcome a string of daunting challenges that run the gamut of engineering specialties: materials, acoustics, electronics, and more.
Can they create a new ultrasound device with the groundbreaking capacity to detect what’s going on inside the microvasculature around the spine? And can they do this with wireless and biocompatible materials, as the concept is to stick that machine on the spinal cord in the manner of a tiny Band-Aid armed with thousands of electrodes? Will they be able to outfit a catheter with a groundbreaking fiber optic array that gives physicians real-time, bedside readings of pressure, temperature, and oxygenation levels deep inside of microenvironments around the spine?
“This is as pioneering as it gets,” Manbachi says. The work that began in an undergraduate educational setting is now in the hands of a multidisciplinary team that includes not just scientists at the Whiting School and the School of Medicine, but also Johns Hopkins’ Applied Physics Laboratory, Columbia University, and a top-of-the-line private technology firm. Former student leader Ainechi is on the team too, from her new perch as a graduate student.
“The way this began is very important to me,” Manbachi says. “The work students do can make it seem like engineering is all about coding and writing scripts and textbook materials. It’s in projects like this that they come to see how this field can actually help people.”
One-Two Punch
The lack of progress in spinal cord injury repair does not reflect a lack of effort. Scores of drugs have entered clinical trials in recent decades. In the early 2000s, stem cell therapies seemed tantalizingly close to making a difference at the bedside. “I don’t mean to be a nihilist. But the fact is, nothing has worked,” says Theodore.
Over time, Theodore developed a theory about that run of failure. The injuries involved in these cases usually involve multiple, interrelated biological events of dizzying complexity. “The cellular cascade that happens in the immediate aftermath of injury is just the beginning,” says Theodore. “After that come secondary injuries—loss of blood flow, loss of oxygen, inflammation, and scar tissue forming.” Those secondary cascades often bring on new levels of paralysis.
“If you’ve got 50 holes in a dike and you stick your finger in one of them, you’re still going to drown,” he says. Therein lies the problem that Theodore brought to the Whiting School five years ago: Can biomedical engineers help to bring that entire flood wall and all of its interrelated holes into focus?
The solution envisioned in the DARPA project involves a pair of devices. One is a catheter; the other is ultrasound. If engineers succeed in making these novel devices work as planned, the one-two punch will open up a wealth of biological data, delivered in real time with an astonishing level of detail.
“We’ll be able to see, in great detail, what’s happening with blood flow and oxygenation in the tiniest microvasculature of the spinal cord,” Manbachi says. Such an instantaneous diagnostic treasure trove will put clinicians in a much stronger position to anticipate and prevent those secondary cascades—and, perhaps, to accomplish more.
A Catheter Like No Other
The “acute” phase after a spinal cord injury—the two weeks or so after decompression surgery—is fraught with risks that are difficult to predict and often impossible to prevent.
Physicians monitor blood pressure very carefully in this period, tinkering frequently to make sure nothing is awry with perfusion and blood flow. During surgery, they also install a lumbar drain that helps maintain healthy fluid levels after the operation.
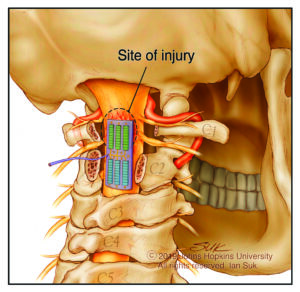
This catheter is what’s being transformed in the first of the one-two punch combination. Work now underway at the Applied Physics Laboratory and the Whiting School aims to turn that catheter into a high-powered diagnostic tool equipped with a trio of fiber optic cables. One would read fluid pressure. Another would track temperature. The third would measure oxygenation.
But the spine is a difficult environment to monitor with current technologies. In brain injury, Theodore notes, surgeons drill a hole in the head and insert a tube in the fluid-filled space of the brain, gauging intracranial pressure. “But the anatomy of the spinal cord is different—its diameter is about the size of your pinkie finger,” says Theodore. “I can’t stick something into the spinal cord without causing injury.” The way things stand now, he adds, surgeons are “up on a tightrope with a blindfold on.”
That blindfold might come off with localized measures of pressure. “The ability to see what’s happening with pressure in blood vessels that are smaller than a human hair would create tremendous opportunities for progress,” he says.
The third data point—oxygenation—presents an especially daunting engineering challenge, says Steve Babin, a physician, electrical engineer, and senior APL researcher. Today, the only way to get an oxygenation number like that is by shipping a sample off to a lab. That leaves physicians operating on information that is at least several hours out of date.
But the only way to measure oxygenation via fiber optics will be through a proxy chemical present in the fluid flowing through the lumbar drain. This notion that fiber optics can be used to find biomarkers continuously is relatively new, but Babin has a record of success, as he is the inventor on a Johns Hopkins patent for a fiber optic sensor that was subsequently shown to measure the compound bilirubin, something that’s of great interest in neonatal intensive care units.
A number of chemicals would be good candidates for the role of oxygenation proxy, but the challenge is finding the right balance between accuracy and feasibility. Different chemicals absorb light in different quantities and at different wavelengths, so their presence can be detected by measuring how much light makes a return trip through the catheter after hitting a retroreflector. This method allows fast, real-time, in situ, and— crucially, in a challenging environment like the spine—safe biomarker measurement.
“That will tell us what the concentration of that chemical is,” Babin says, “but we need to find the right chemical, the one that has wavelengths that are in an area that we can access, technologically.” The winning candidate from this pool of chemicals has yet to emerge.
Other technical hurdles loom on the catheter horizon. The logistics of fiber optics mean this souped-up catheter can’t be inserted with a traditional needle. Medical device expert and microelectronics process engineer George Coles of the APL team is developing a brand-new way of inserting the catheter.
Another question surrounds the thickness of the fiber optic cables: How much will the cables inhibit fluid making its way through the drain?
“Nick sometimes needs all the capacity that’s in the drain,” Manbachi says. “We’re still figuring that out too, experimenting with different elasticities.”
Biomedical engineer Francesco Tenore is confident the APL team is on the road to meeting all these challenges. “When physicians are able to see these results coming in continuously in real time, that’s going to be a medical first,” he says. “It’s going to provide them with immediate feedback on their therapies, so that they can alter and modify it.”
Bringing Ultrasound to a New Environment
Ultrasound made its first splash in medicine during the 1970s, once the invention of rudimentary scanners gave physicians a way to visualize the information available through high-frequency sound waves. In short order, it became an everyday tool across scores of specialties. Think fetal imaging in obstetrics, echocardiograms in cardiology, and bone sonometry in orthopedics.
Manbachi’s team is looking to push ultrasound into uncharted territory by imaging the microvasculature around the spine. The largest spinal arteries are tiny as can be, measuring in the neighborhood of 200 microns. Countless even smaller tributaries run this way and that from such main lines. In tight quarters like this, blood moves more slowly than it might through an artery like the carotid, which measures about 1 centimeter.
“A typical ultrasound probe might run between 5 and 10 megahertz,” Manbachi says. “We’re looking at running this between 20 and 33 megahertz, because we’re going to need to really push those limits to make sure we can see what we need to see.”
The environment is not the only groundbreaking aspect here. The device will need to win regulatory approval as biocompatible, since the plan is for surgeons to attach it to the spine during decompression surgery. (The implanted device will not use bulky batteries. Instead, it will be wirelessly powered from a nearby relay station, outside of the body.)
Making matters more complicated is the human spine itself. It might look rigid on a classroom skeleton, but inside a living body it’s constantly pulsating along its entire length. The rates vary, not just among different people, but within a single individual, especially one who has been through recent injuries and surgeries, and is probably still adjusting to new medications.
“This is going to be a big acoustic engineering challenge,” Manbachi says. “How do you keep your aim on the right focal point when the whole spine is pulsing like that?”
No ultrasound machine has ever operated amid the distractions and complications of a living environment like this. How will the flow of spinal fluid affect sound waves? What will happen to those waves as that flow fluctuates because physicians are adjusting the amount of fluids being drained through a catheter?
The broader scientific team tackling these challenges includes radiologists, neurosurgeons, and neurologists in addition to biomedical engineers. A multidisciplinary subteam is housed at the private-sector firm, Sonic Concepts, that will build the actual devices, which will measure just 1 centimeter by 2 centimeters while armed with a full array of thousands of ultrasound transducers.
The end goal is to have a device ready for human testing on the ambitious five-year timeline of the grant.
A Therapeutic Tool
The potential this device holds for improving patient outcomes is enormous. Armed with detailed, highly localized three-dimensional imaging, physicians will be in a much stronger position to gauge the risk of secondary injuries associated with scarring, inflammation, and other post-injury phenomena. These biological “cascades” have devastating long-term impacts, turning a patient who may initially have lost the use of an arm or a leg into a paraplegic or quadriplegic.
Paralysis sets in among spinal injury patients when the body loses its knack for autoregulation, no longer able to figure out how much blood should flow through this artery and when this or that electrical impulse should fire.
The ultrasound device envisioned in this project will do more than help physicians monitor these risks; it will also operate as a therapeutic tool. When signs of neurogenerative failure appear, physicians will be able to step in with “focused ultrasound.” Manbachi compares the neuromodulation nudge this delivers at the cellular level to a gentle tap on the cheek to a person who is nodding off to sleep.
In addition, recent laboratory work has explored the use of electrodes to drive electrical stimulation along the spine, which puts electric stimulation therapy on the table as well. That approach may or may not live up to its potential—only time will tell. For now, Theodore prefers to keep his eye on the prize of incremental improvements.
“If we can change just those secondary outcomes, saving something like arm mobility, it will make a big difference for patients.” he says. “We’d all love to find a way to ‘cure’ spinal cord injury. But our first steps toward good therapeutics will be baby steps, and we need to remember that those are going to help patients too.”
Everyone involved in this project senses that the completion of the five-year journey through DARPA is more likely to mark a beginning than an ending. Manbachi views the destination here as a “system of systems” where ultrasound and fiber optics are linked and communicating, and data gathering is happening on the machine-learning level.
APL’s Tenore is looking forward to the day when new patients can benefit from readings gleaned in thousands of prior patients. “This opens up the possibilities of bringing machine-learning algorithms to bear here,” he said. “That, in turn, opens the way to personalized medicine, as it will become quickly apparent in many cases how any given patient differs from ‘the norm.’”
As the new devices get up and running, specialists in other fields are bound to take note. “Spinal cord injury is our focus here, but it’s not limited to that down the road,” says Tenore. “If we demonstrate that we can use these technologies to track these biological changes in real time, that opens up a lot of other possibilities.”
Theodore agrees: “The ability to make these measurements in a microenvironment with implantable devices might end up being used in the brain or on the liver—who knows where else? We’re working at the tip of a very big iceberg here. I really believe that this has the potential to transform clinical medicine.”
Says Manbachi: “I’ve always dreamed of becoming an engineer whose work actually helps patients at the bedside. I feel like this grant is giving me an opportunity for that dream to come true.”






