Current Projects:
Room Temperature Electrochemical Synthesis of Ordered Intermetallic Materials
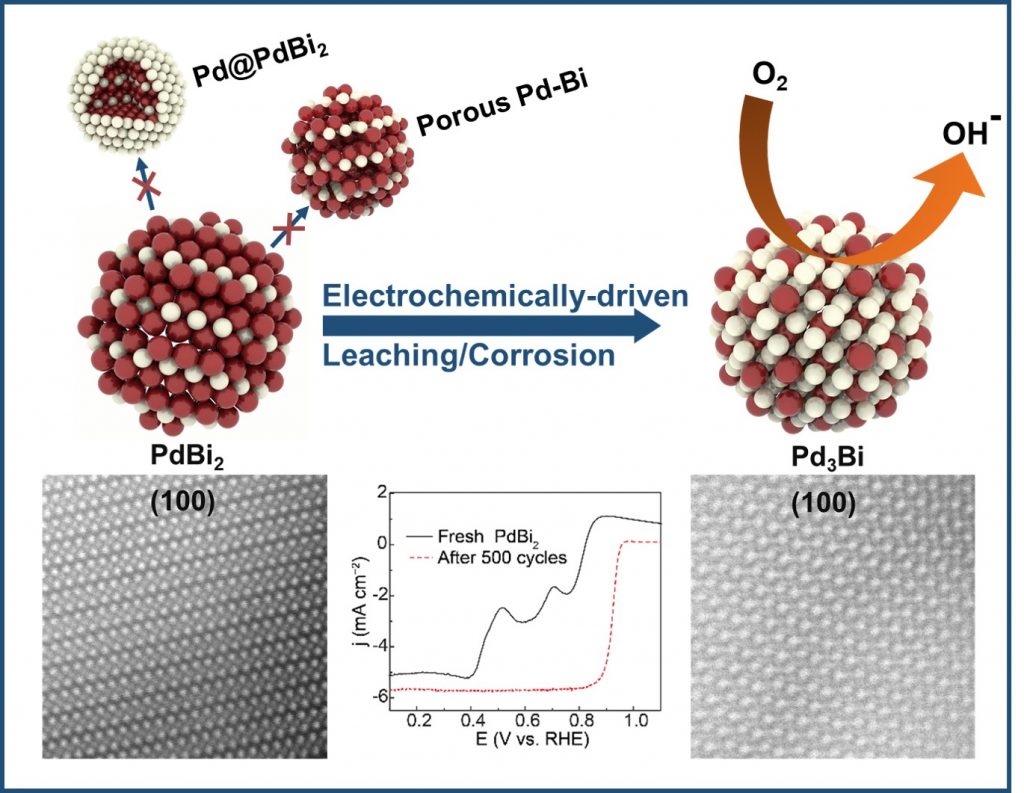
Ordered intermetallic compounds (OICs), alloys with long range atomic ordering, exhibit important and useful properties for catalysis, electronics, corrosion/sintering resistance and other applications and are often superior to solid-solution alloys when in a nanostructured form. Despite decades of intense research, OIC nanoparticles have failed to replace conventional nanomaterials due to (1) lack of low-temperature synthetic methods which can overcome slow solid-state diffusion rates which inhibits atomic ordering, (2) inability to tune composition and phase to optimize the desired application, and (3) lack of fundamental understanding needed for progress on these issues. The Hall group is currently developing electrochemical methods (e.g. electrochemical dealloying, electrodeposition) to prepare nanostructured OICs at room temperature and atmospheric pressure. Our experimental program has laid the foundation for a fundamentally new approach for the synthesis of nanostructured OICs at room temperature. We will uncover meaningful trends that will enable us to link our insights to produce materials with tailored compositions, permitting the design of high-performance nanomaterials which are difficult to access by conventional methods.
Electrochemically Mediated Renewable Energy Conversion

The Hall group investigates chemical reactions catalyzed by solid surfaces to address problems in renewable energy storage and utilization. We design ordered intermetallic materials interrogate their performance for mediating electrochemical reactions. Ordered intermetallic materials are advantageous over disordered alloys because they usually exhibit improved catalytic performance and stability. We also interrogate the adsorption dynamics of the metal-solution interface by Surface Enhanced Infrared Absorption Spectroscopy (SEIRAS) to understand how material structure and solvent play a role in controlling catalytic performance. We are interested in probing reactions such as fuel cell catalysis, electrochemical CO2 reduction, and electro-organic chemistry.
